 
A free settling particle's motion is not affected by that of other particles, the vessel's walls, or turbulent currents. The settling rate of a particle is defined in terms of "free" versus "hindered" settling. Most waste treatment systems employ a gravity separation step for suspended particle or oil removal. Essentially, biological waste treatment is this conversion of soluble contaminants to insoluble forms. Ordinarily, water-soluble contaminants are chemically converted to an insoluble form to allow removal by physical methods. Various physical methods may be used for the removal of wastewater contaminants that are insoluble in water, such as suspended solids, oil, and grease. Large quantities of discharged salts necessitate expensive removal by downstream industries using the receiving stream for boiler makeup water. Metals can cause product quality problems for industrial users. Aquatic life is affected by the wide swings in pH as well as the destruction of bicarbonate alkalinity levels.Ĭertain metals are toxic and affect industrial, agricultural, and municipal users of the water source. The natural buffering system of a water source is exhausted by the discharge of acids and alkalies. Some solids can coat fish gills and cause suffocation. Deprived of a light source, photosynthetic organisms die. Suspended solids increase the turbidity of the water, thereby inhibiting light transmittance. 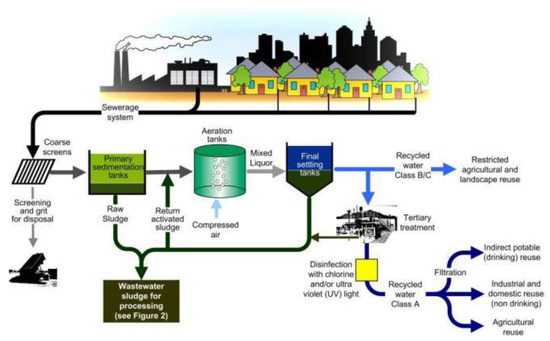
Settled solids cover the bottom-dwelling organisms, causing disruptions in population and building a reservoir of oxygen-consuming materials. Solids discharged with a waste stream may settle immediately at the discharge point or may remain suspended in the water. During the day, algae produce oxygen through photosynthesis, but at night they consume oxygen. Phosphorus is a concern because of algae blooms that occur in surface waters due to its presence. Certain organisms use nitrogen as a food source and consume oxygen. However, nitrogen compounds can have the same effect on a water source as carbon-containing organic compounds. Nitrogen and phosphorus are essential to the growth of plants and other organisms. Organic compounds are normally measured as chemical oxygen demand (COD) or biochemical oxygen demand (BOD). Under extreme conditions, when the dissolved oxygen concentration reaches zero, the water may turn black and produce foul odors, such as the "rotten egg" smell of hydrogen sulfide. When the organic load causes oxygen consumption to exceed this resupply, the dissolved oxygen level drops, leading to the death of fish and other aquatic life. 
Atmospheric oxygen can replenish the dissolved oxygen supply, but only at a slow rate. In a biochemical reaction, dissolved oxygen is consumed as the end products of water and carbon dioxide are formed. 
Organisms in the water use the organic matter as a food source. The amount of organic material that can be discharged safely is defined by the effect of the material on the dissolved oxygen level in the water. This practice also reduces water consumption. Wastewater can be recycled for reuse in plant processes to reduce disposal requirements (Figure 37-2). Special processes are required for the removal of certain pollutants, such as phosphorus or heavy metals. In most cases, treatment is required for both suspended and dissolved contaminants. Wastewater treatment plants are designed to convert liquid wastes into an acceptable final effluent and to dispose of solids removed or generated during the process. This assimilative capacity varies with the type and amount of pollutant. Consideration is given to the feasibility of removing a pollutant, as well as the natural assimilative capacity of the receiving stream. The permitted quantities are designed to ensure that other users of the water will have a source that meets their needs, whether these needs are for municipal water supply, industrial or agricultural uses, or fishing and recreation. Discharge permits, issued under the National Pollutant Discharge Elimination System (NPDES), regulate the amount of pollutants that an industry can return to the water source. Improvements in determining the effects of industrial waste discharges have led to the adoption of stringent environmental laws, which define the degree of treatment necessary to protect water quality. Because some of this water becomes contaminated, it requires treatment before discharge (Figure 37-1). Many industries use large volumes of water in their manufacturing operations. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
